Cell Image Analysis
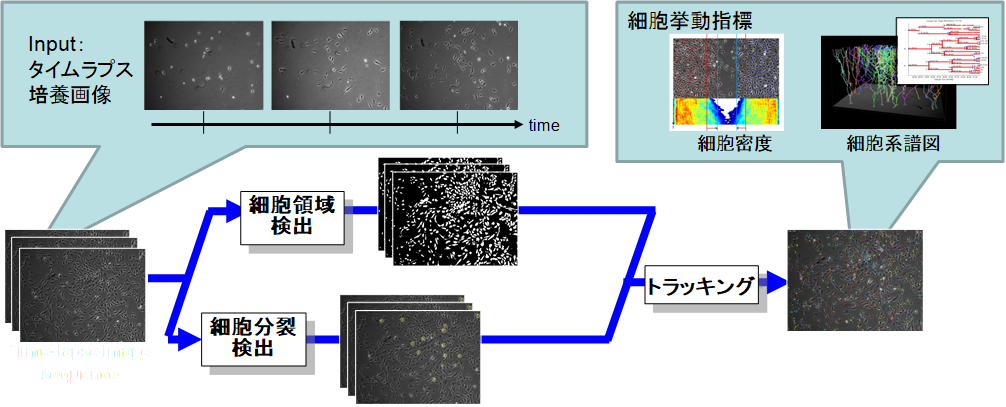
Advances in optics and imaging systems have enabled biologists to visualize a living specimen's dynamic pro-cesses by time-lapse microscopy images. However, the image data recorded during even a single experiment may consist of hundreds of objects over thousands of images, which makes manual inspection a tedious and inaccurate option. We addresses the need for computational tools and community resources that can enable the quantitative system-level biology automatically. The domain of our current interests are cell image analysis algorithms including microscopy image restoration, cell mitotic event detection and cell tracking by data association, which are integrated into an online automated system.
Medical Image Analysis
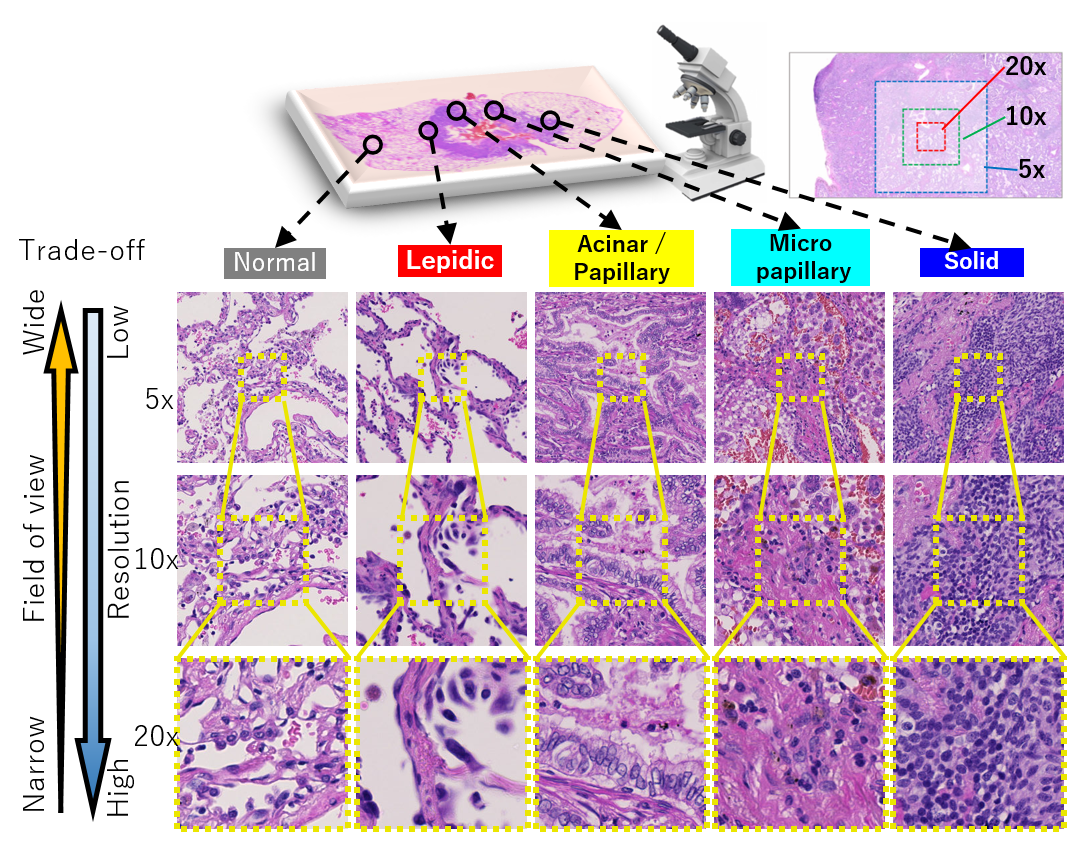
In order to improve the quality and safety of medical care, sophistication, efficiency, and equalization, a foundation for collecting and using medical big data represented by medical images using ICT is required. In this research, we are developing new technologies using the latest image analysis technology, such as deep learning for large amounts of medical image data. In addition to research and development that uses public datasets simply set up with tasks, we will collaborate with doctors and discuss what tasks we should work on, including how to create training data. Current our main targets are pathological images and endoscopic images.
Photoacoustic Imaging and Blood Vessel Analysis (ImPACT Project)
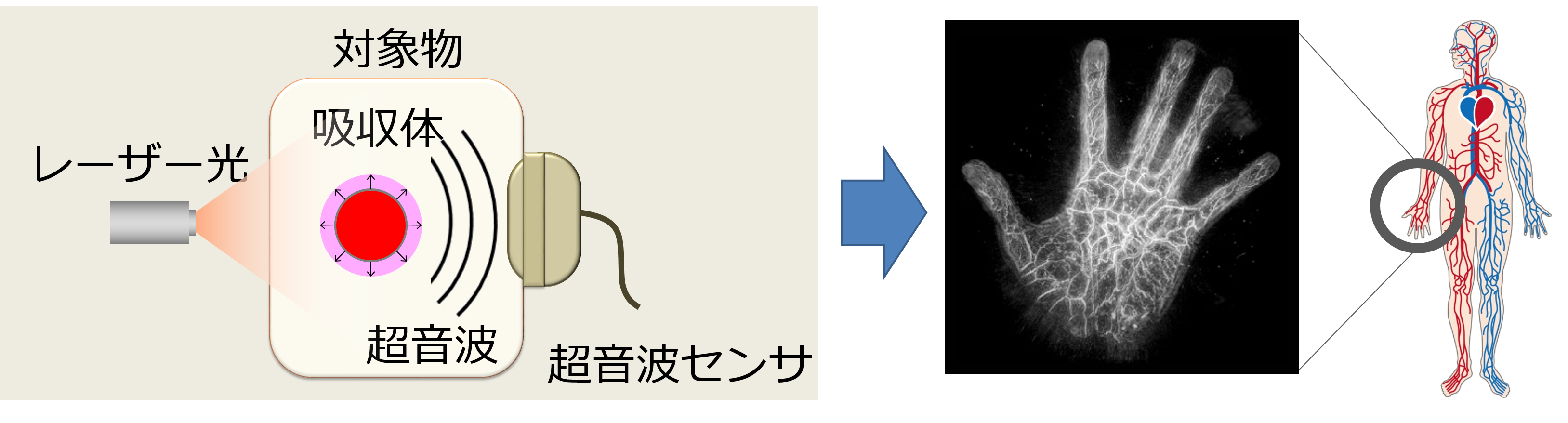
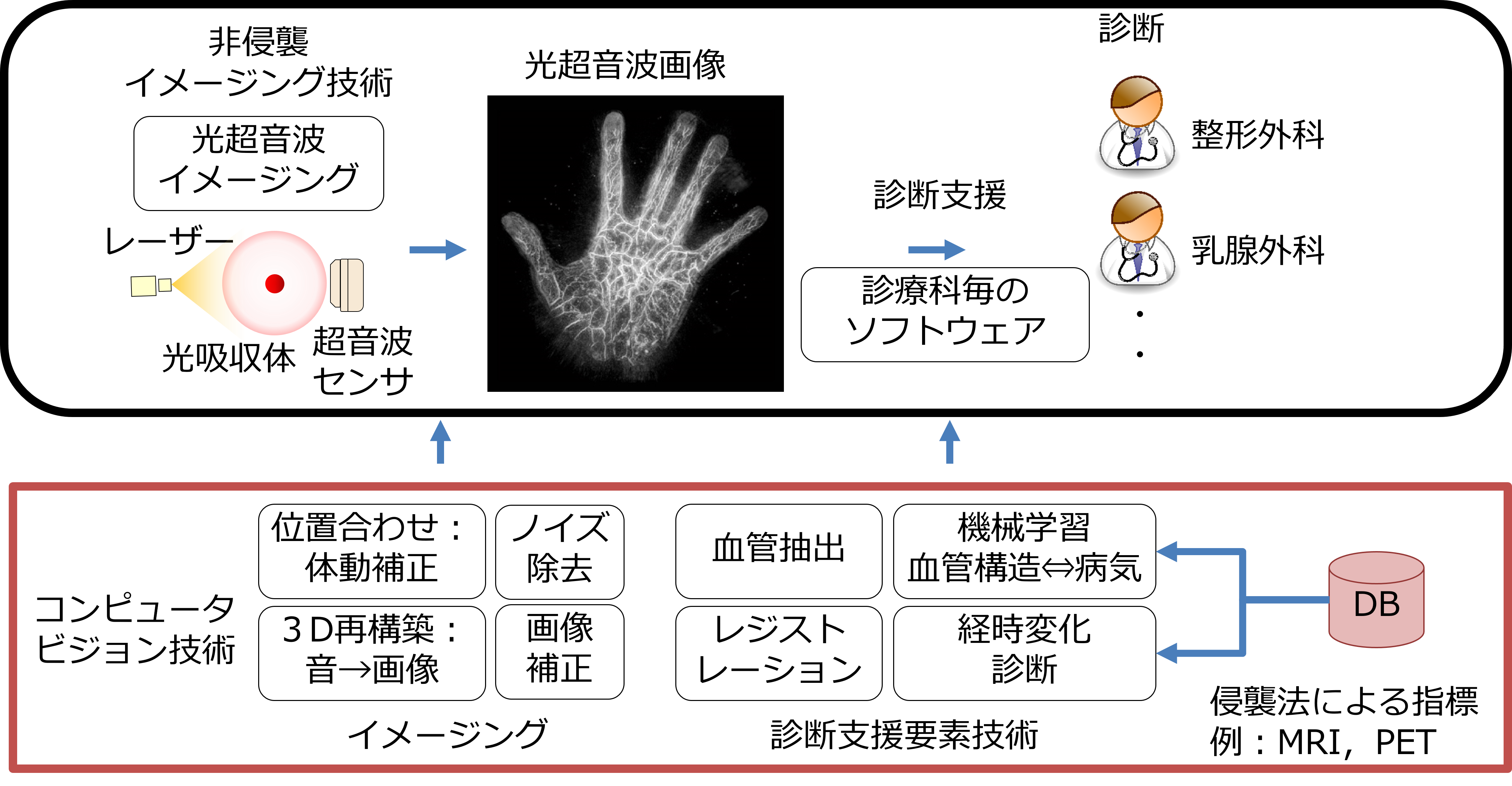
With the arrival of super-aging society, there is increasing demand for technical support to enable people to continue working while preserving their health and beauty. NII has participated in ImPACT to realize an early diagnosis of disease, and inspection of the internal structure, with advances in photo-acoustic imaging, which performs real-time 3D visualization of changes in properties and functions inside human bodies and substances, non-invasively and non-destructively. The photo-acoustic system is a promising new technology that integrates state-of-the-art laser and ultrasound technologies, where 3D structures of objects can be reconstructed by sensing emitted ultrasound from the objects that absorb near-infrared irradiation. It enables to image the state of the human body and objects whose insides are not visible, non-invasively and non-destructively. In this research, we develop computer-vision technologies to obtain clear images and extract bio-image features to support a diagnosis. For example, we proposed a registration method to generate high-quality 3D volumes in which vessels become clearly visible by aligning shot-volumes that are misaligned by body motions. We are also developing a technology that automatically models vascular structures, which helps in understanding blood vessel conditions strongly related to illnesses.
